ARcare® 94220
Product Applications
The typical use for this product is in the securing wearable devices for medium term wear (3-7 days). However, it may also be used in other skin bonding applications including in wound care or dressing attachment.
Features and Benefits
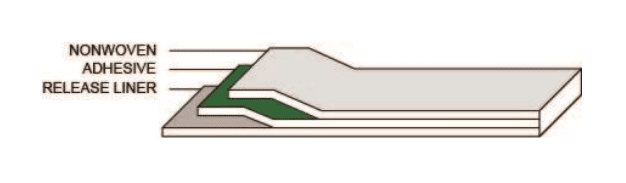
Product Construction
| Typical Values* | Description | |
|---|---|---|
| Carrier thickness | 11 mil | 279 μm | Non-woven PET carrier |
| Adhesive thickness | 2.6 mil | 66 μm | MA-183 medical grade adhesive |
| Liner thickness | 3.2 mil | 81 μm | White paper liner |
| Total thickness | 13.6 mil | 345 μm | (Excluding liner) |
*All stated values are nominal and should only be used as a guide for selection. They are not specifications.
Safety Data
The base polymer of MA-183 has passed cytotoxicity, skin irritation and skin sensitization testing conducted by an independent laboratory. Additional safety information is available upon request.
Technical Properties
| Attribute* | Typical Values* | Test Method* |
|---|---|---|
| Peel adhesion on stainless steel | 55 oz/in | 15 N/25 mm | 90 degrees, 12 ipm, 15 min. dwell |
| Release of polyester liner | 70 g/2” | 90 degrees, 12 ipm |
| Moisture vapor transmission rate | > 500 g/m2/day | Payne Cup Method, ART 4047 |
| Recommended storage of unconverted product | 70°F ± 20°F | 21°C ± 11°C 50% ± 20% RH | 50% ± 20% RH |
|
| Shelf life of unconverted product | Not to exceed one year from date of manufacture |
*All stated values are nominal and should only be used as a guide for selection. They are not specifications.